Abstract: Urothelial carcinoma (UC) is the second most frequent urogenital malignancy with high incidence in the United States and Europe. Despite poor prognosis, new treatments have emerged with great efficacy and safety such as immune checkpoint inhibitors. Durvalumab, an anti Programmed Death Ligand 1, has been given breakthrough in UC in 2017 in patients who have disease progression during or following platinum-containing chemotherapy or who have disease progression
Keywords: immunotherapy, PD-L1 inhibitor, durvalumab, urothelial carcinoma, bladder cancer
INTRODUCTION
Urothelial carcinoma (UC) also known as transitional cell carcinoma is the ninth most common cancer worldwide with 430,000 new cases and 165,000 deaths in 2012.1,2 In Europe, the incidence is high with 151,297 new cases in 2012 and 52,411 deaths.2 Despite the poor prognosis of the disease and the 30% rate of muscle-invasive carcinoma at diagnosis, treatment has remained the same for decades. The cornerstone of metastatic or unresectable disease was platinum-based chemotherapy. The combination of methotrexate, vinblastine, doxorubicin, and cisplatin (M-VAC) regimen has been considered for first-line therapy in fit patients for 30 years with objective response rates (ORR) around 50%–70%. The association of gemcitabine and cisplatin (GC) has been compared to M-VAC in a Phase III study, showing no improvement in overall survival (OS; M-VAC 14.8 months vs GC 13.8 months), but the study was not powered enough to show equivalence between the two regimens. However, the better safety profile led to an approval of GC as an equivalent of M-VAC.3 These chemotherapy regimens are tailored for fit patients with good performance status (PS) and adequate renal function. However, 25%–50% of patients with UC are not eligible for cisplatin-based therapies because of age-related comorbidities and epidemiology of the disease.4,5
UCs are immunogenic tumors with a high mutational load that can be targeted by immunotherapy.6,7Moreover, intravesical Bacille Calmette–Guerin, an immunotherapy, has been used since 1990 to reduce recurrence in nonmuscle-invasive bladder cancer.8
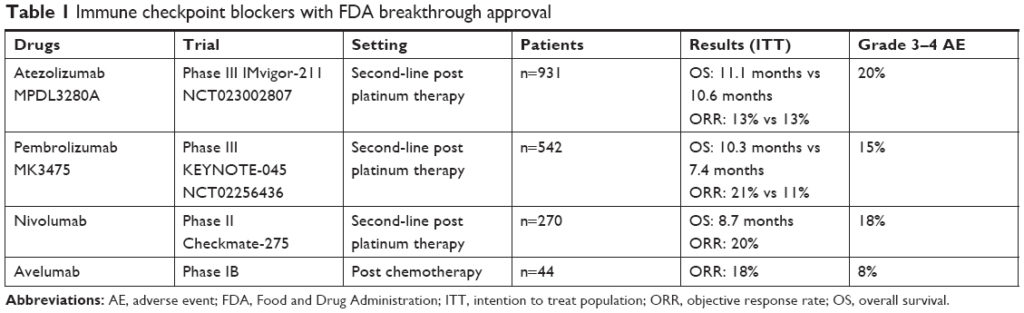
Several immune checkpoint blockers have been given breakthrough in UC since 2016 with promising results (Table 1).9–12 In this review, we will focus on the use of durvalumab, an anti Programmed Death Ligand 1 (PD-L1) in UC.
READ FULL ARTICLE
![]() From Dovepress
From Dovepress
